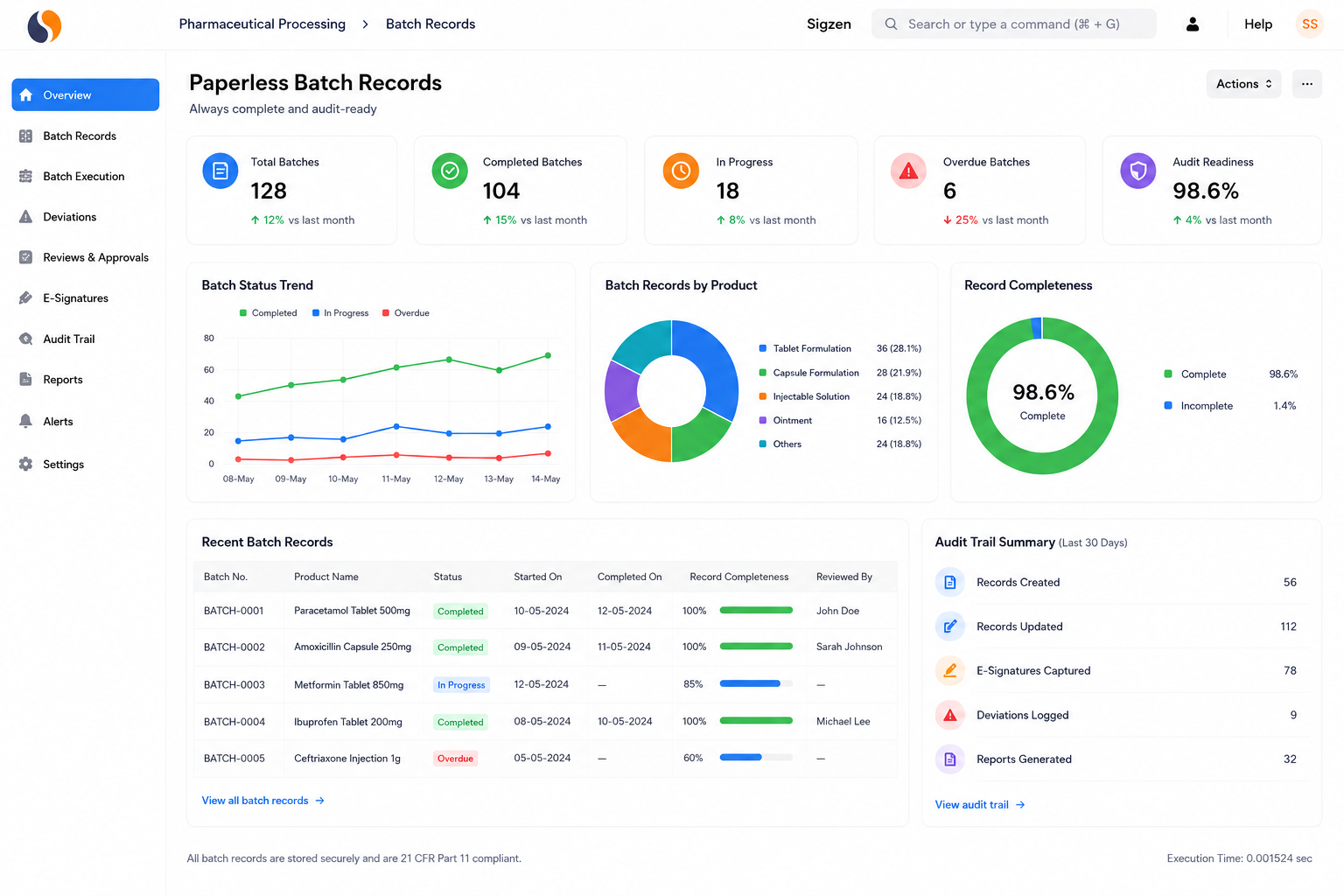
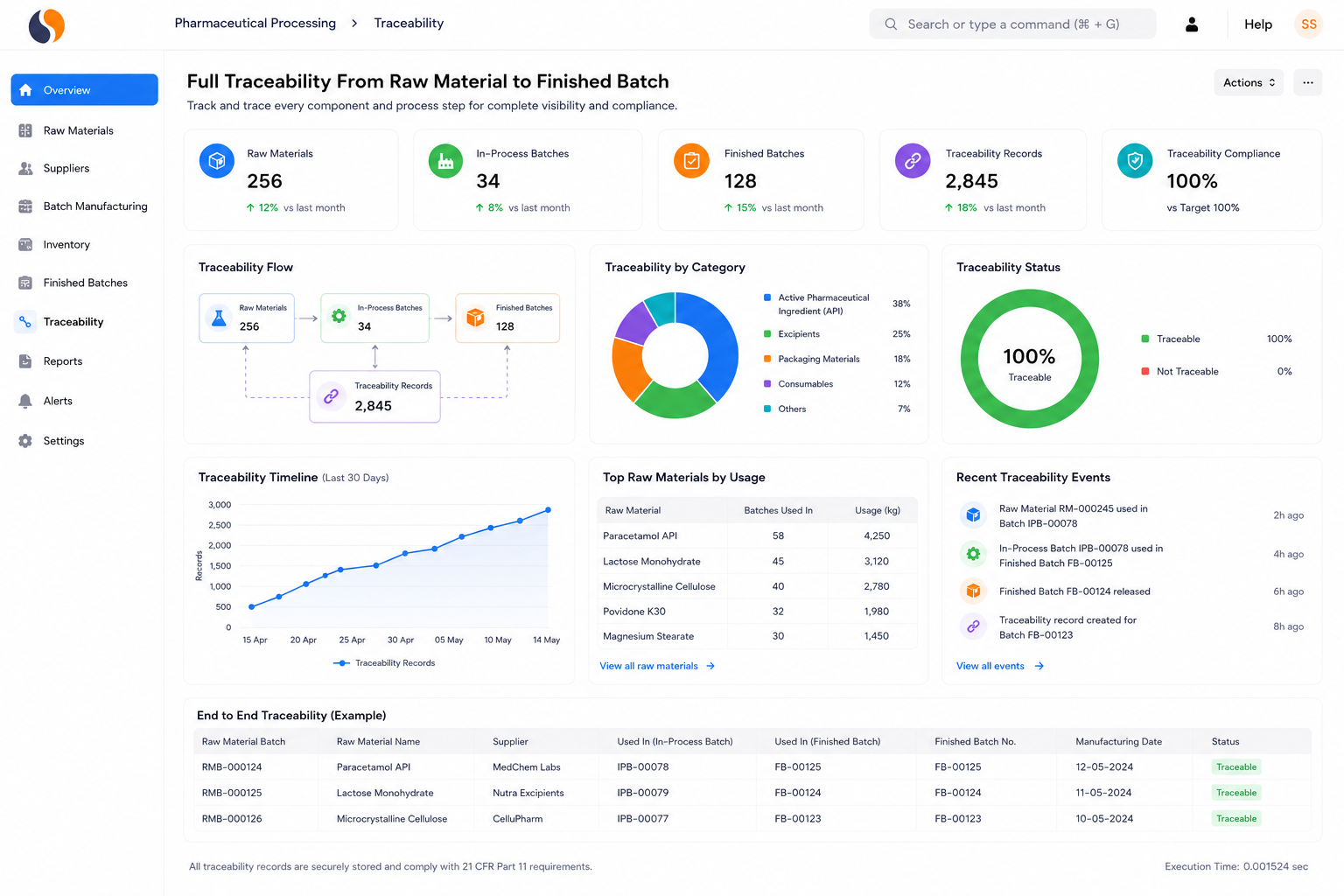
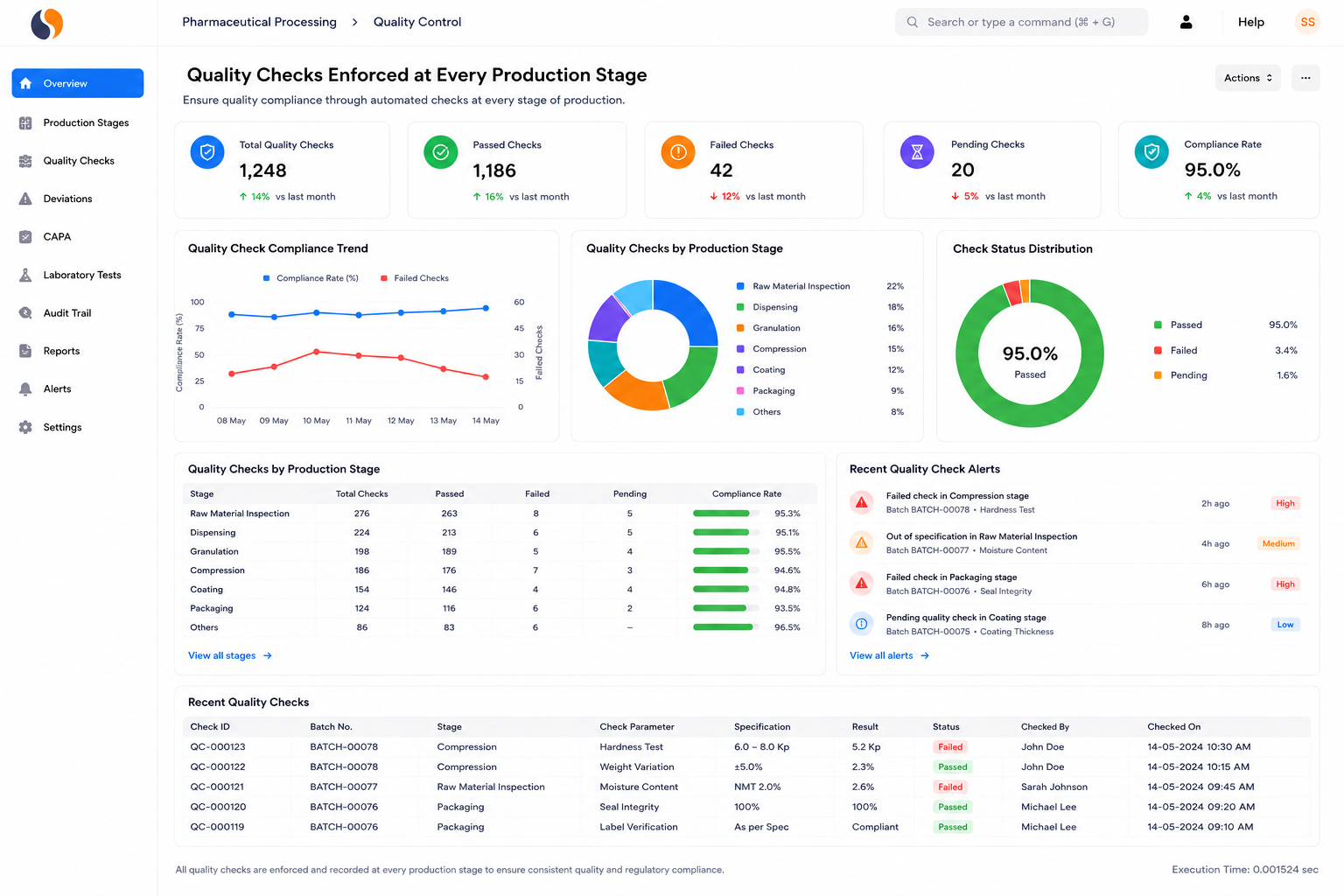
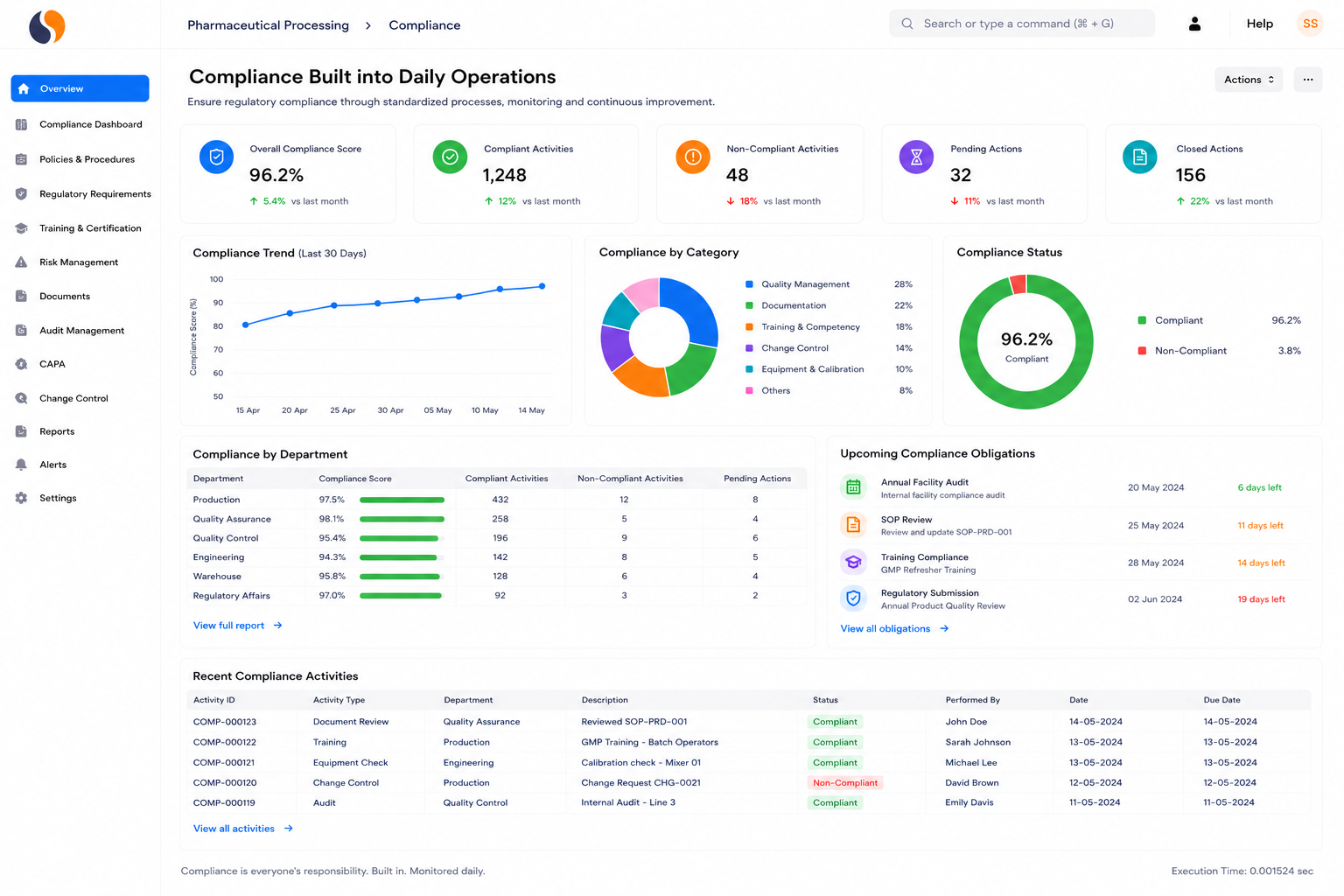
Managing pharmaceutical operations today means dealing with strict audits, batch risks, and scattered data across systems. Whether you manufacture APIs, intermediates, formulations, medical devices, or biotech products, these challenges slow decisions and increase compliance pressure. Sigzen Pharma ERPNext brings production, quality, and compliance into one system.
Pharmaceutical ERPNext Solution - one system for all pharma operations

Explore Our Industry-specific pharma ERP solutions
API Manufacturing
Bulk drug manufacturers and API producers working with complex chemical processes. Managing multi-step synthesis, yield tracking, and regulatory documentation like 21 CFR Part 11 records can become difficult quickly. Get full lot traceability from raw material receipt to finished API dispatch.
Pharma Intermediates
Manufacturers producing chemical intermediates used in API synthesis. Handling multiple production stages with stage-wise yield tracking and mandatory quality checks creates operational delays. Gain stage-by-stage visibility with automatic yield variance alerts.
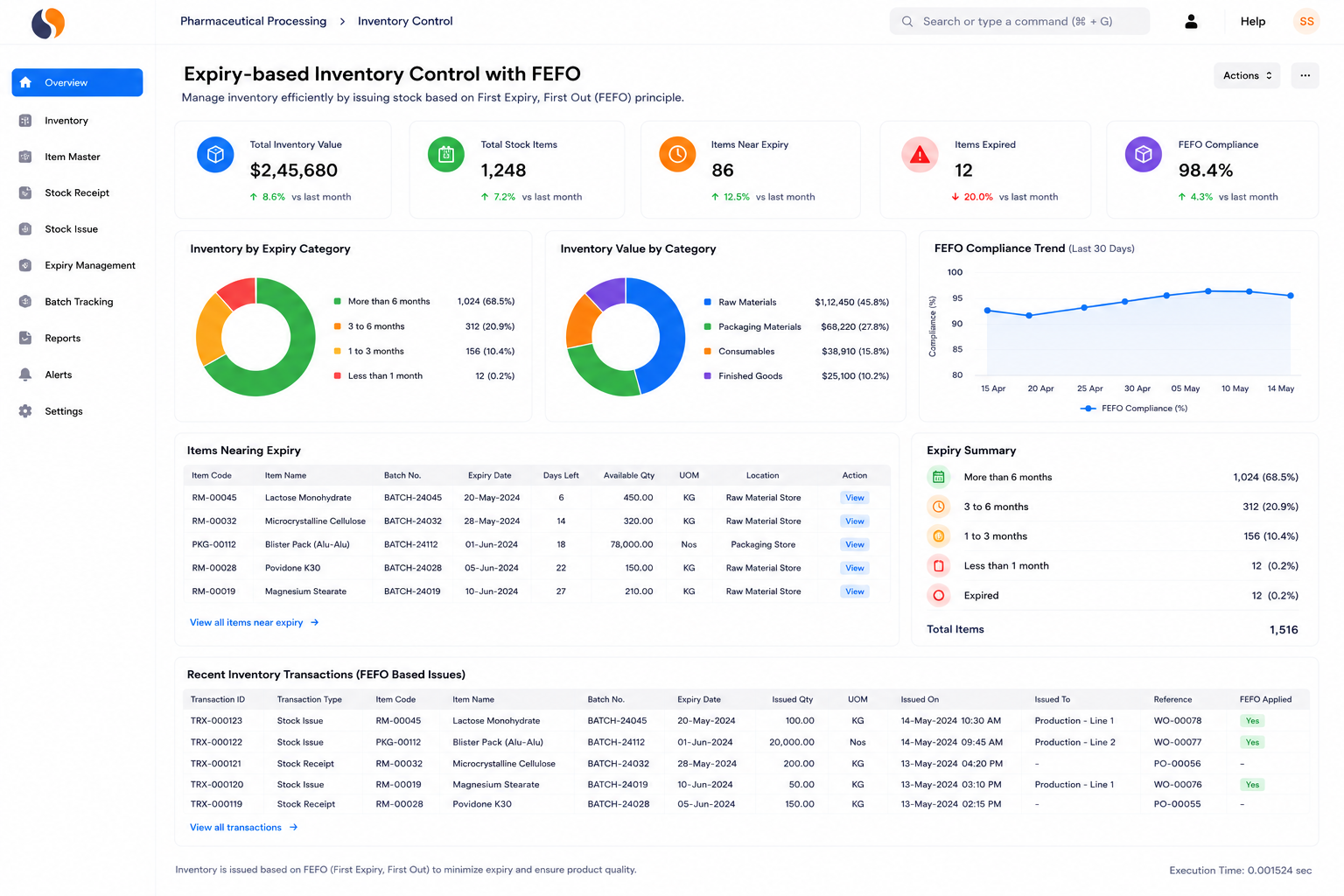
Pharma Formulations
Tablet, capsule, syrup, and injectable manufacturers managing high product variety. Controlling hundreds of formulas, ensuring Schedule M compliance, and handling expiry-based dispatch is difficult on spreadsheets. Achieve formula-to-dispatch traceability with built-in audit records.
Medical Devices & Diagnostics
Manufacturers of devices, kits, and surgical instruments. Managing MDR compliance, serial number traceability, and calibration records across equipment is time-consuming without a system. Maintain device-level traceability with audit-ready documentation.
Biotechnology & Research
Biotech firms and research labs handling experimental and commercial processes. Tracking research costs, lab consumables, and biological batch records alongside regulatory submissions creates complexity. Connect lab-to-production workflows with full traceability and cost tracking.
Why pharma companies need ERP today
India’s pharmaceutical industry is facing tighter regulatory checks while scaling exports rapidly. CDSCO enforcement, WHO-GMP audits, and FDA scrutiny are increasing.
Key Features and Benefits of ERPNext for Pharmaceuticals
Schedule a free ERP consultation for your business
See how ERPNext can improve your day-to-day operations, from production and inventory to reporting and control. Get a practical walkthrough based on how your business actually runs.
Frequently Asked Questions
Which ERP is best for API manufacturing companies?
API manufacturers need pharmaceutical ERP software that supports multi-step batch processes, yield tracking, and strict regulatory compliance. A system with full lot of traceability and digital batch records is essential for audits and exports. For API-specific workflows, See our API Manufacturing ERP Solution.
How do I choose the right pharma ERP for my manufacturing type?
The right pharmaceutical ERP software depends on your production process – API, intermediates, formulations, devices, or biotech. Each segment has different compliance and workflow needs. The best approach is to review a solution built specifically for your segment and evaluate how it matches your operations.
Which ERP system works best for pharma formulations manufacturing?
Formulation companies need strong BOM management, batch traceability, and expiry-based inventory control. Pharmaceutical ERP software should also support Schedule M for compliance and distributor management. For formulation-specific processes, explore our Pharma Formulations ERP Solution page.
Can one ERP system handle multiple pharma business segments?
Yes, a well-configured pharmaceutical ERP software can handle multiple segments under one system. However, each segment – API, formulations, or devices, requires tailored workflows and controls. It is important to ensure that the system supports your primary business first.
How does ERP help medical devices and diagnostics manufacturers?
Medical device companies require device-level traceability, calibration records, and MDR compliance tracking. Pharmaceutical ERP software adapted for devices ensures serial number tracking and audit-ready documentation. For this segment, See our Medical Devices & Diagnostics ERP Solution.