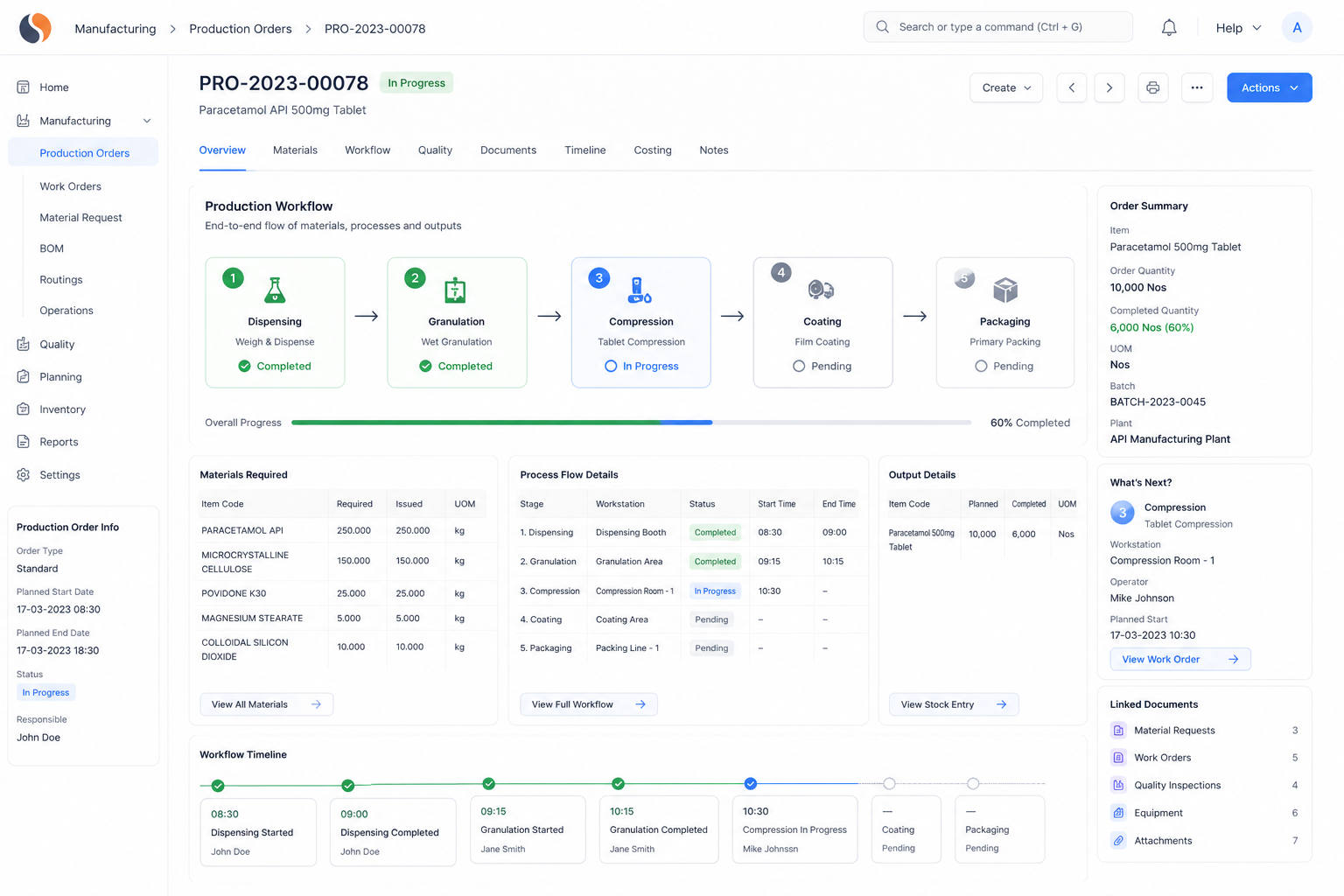
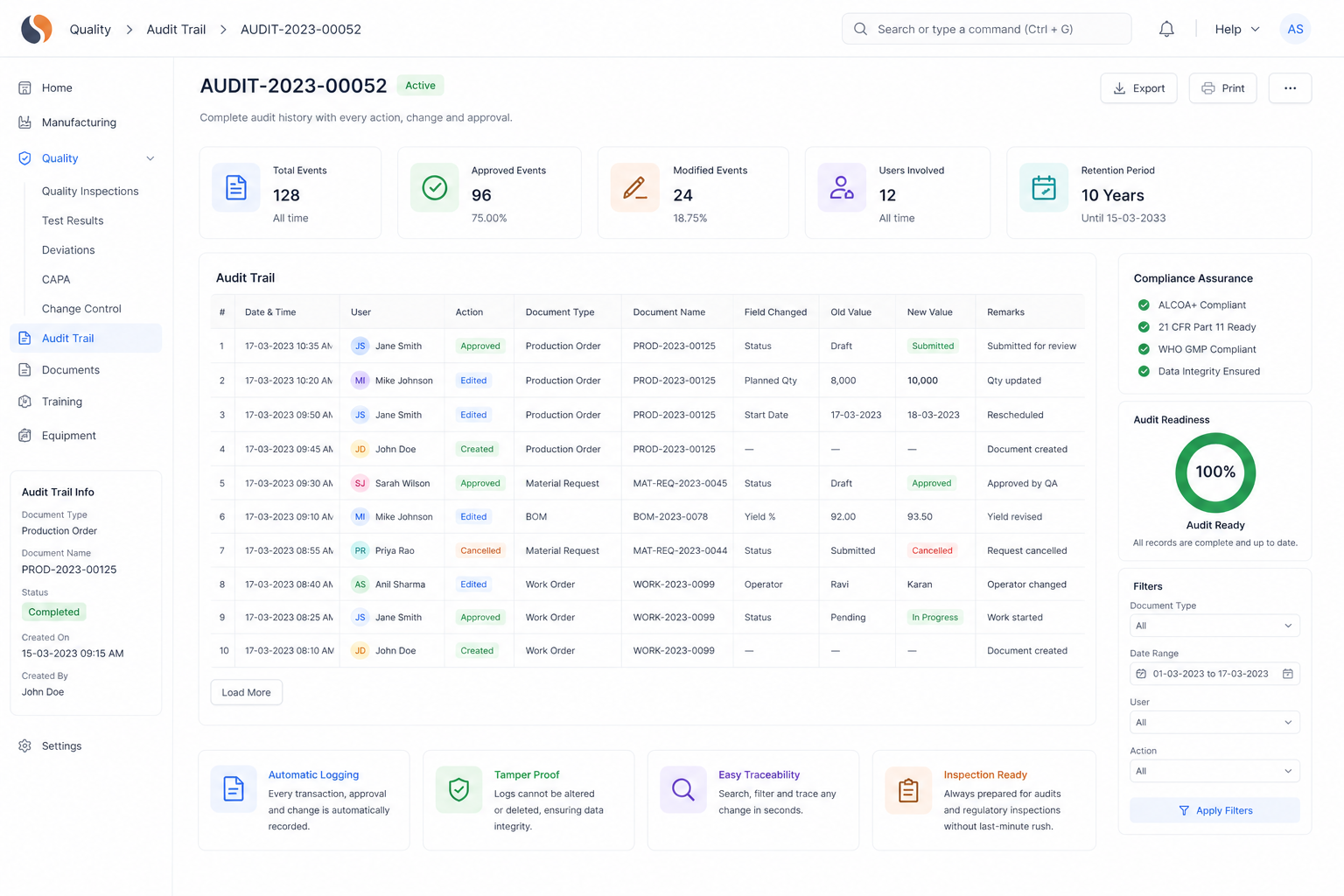
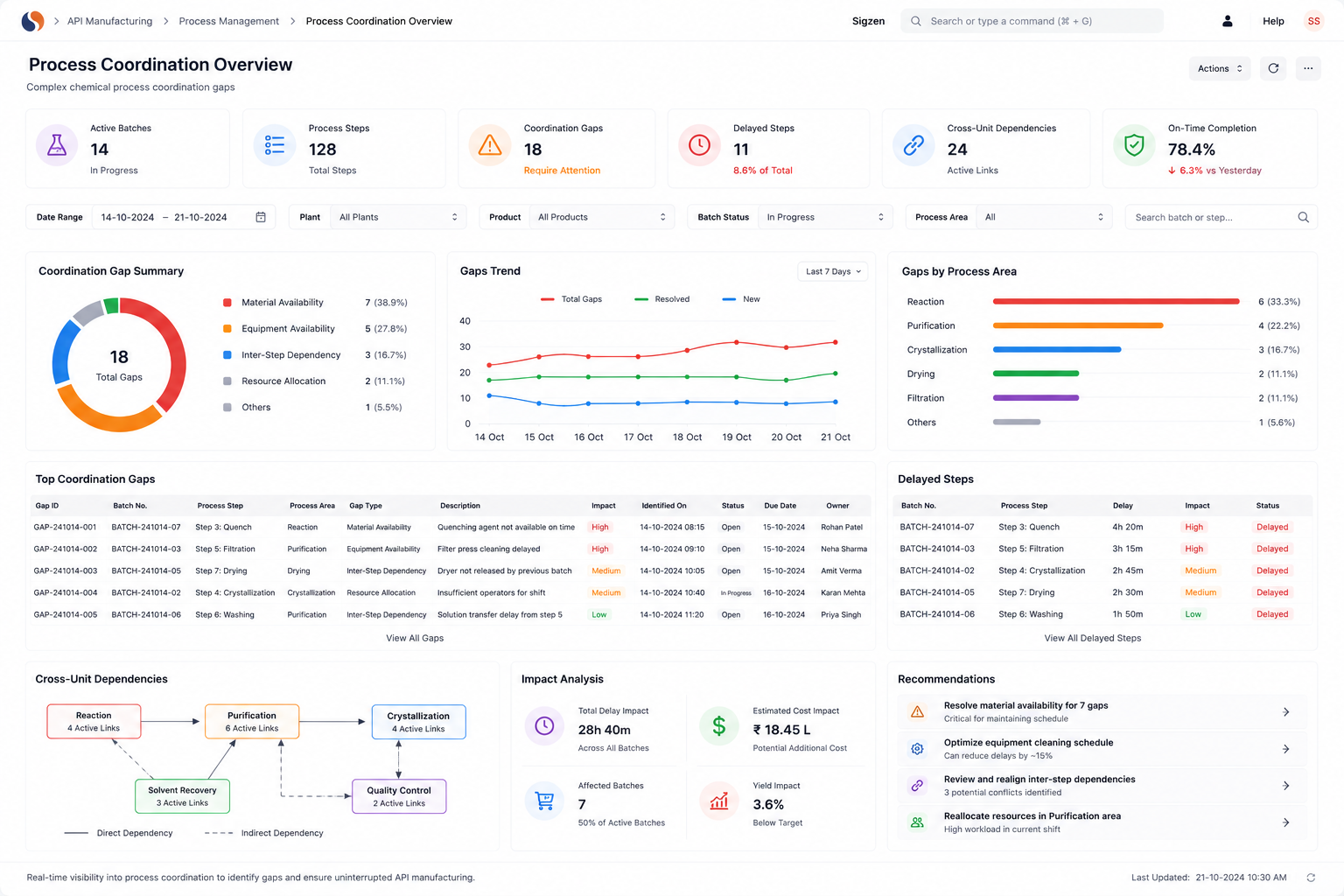
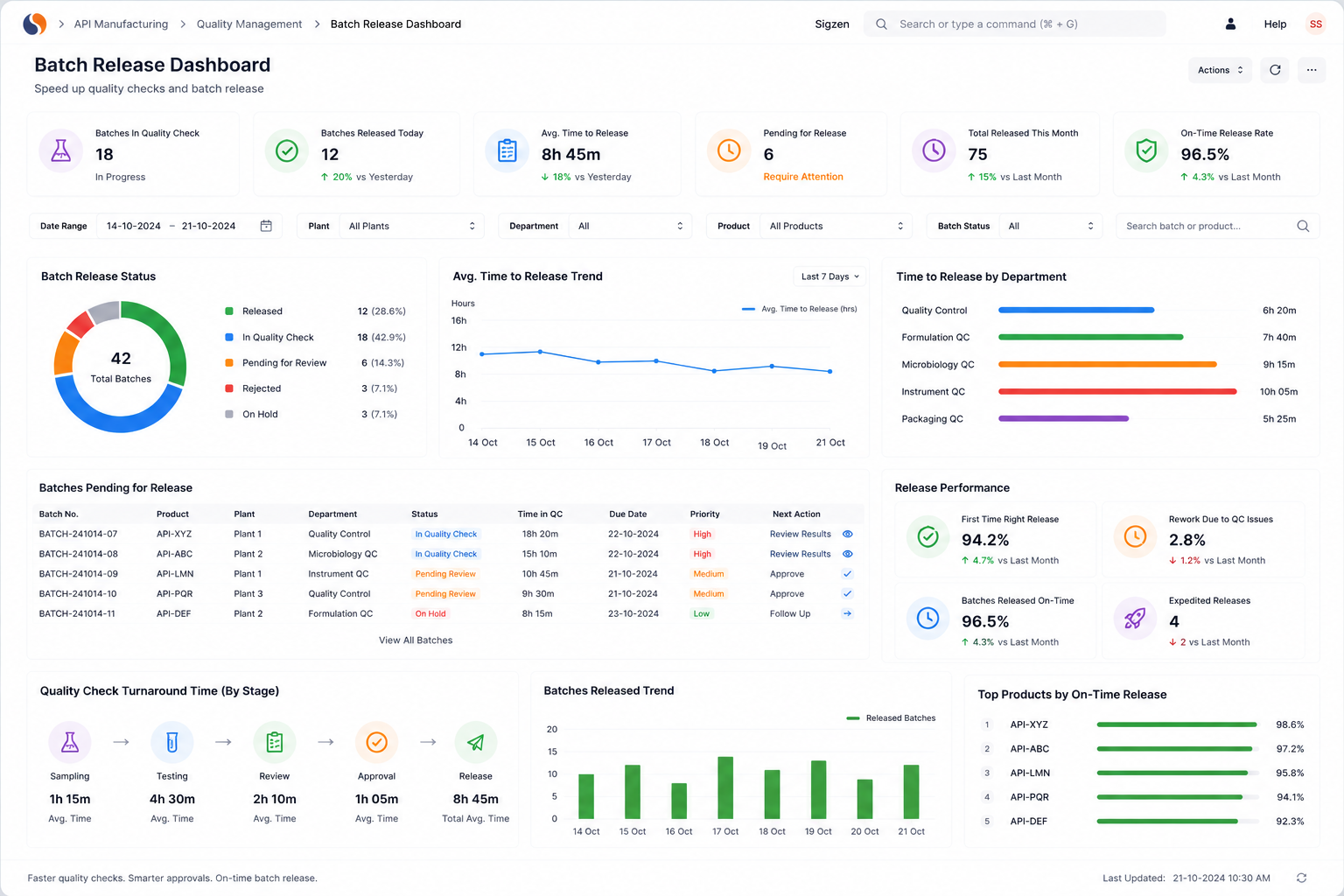
API ERP, API Manufacturing Software, Global Pharma Compliance challenges slow your operations with batch trace gaps, audit stress, and inconsistent yields. With ERPNext implemented by Sigzen Technologies, you gain real-time batch visibility and audit-ready records.
ERP Solutions for API Manufacturing: Active Pharmaceutical Ingredients

API manufacturing software challenges
Key Features and Benefits of API ERP features
Built for compliance and audit readiness
Works with the tools your team already uses
API manufacturing depends on multiple systems working together. Integrations ensure your data flows without manual duplication or delays.
Already using a tool not listed here? Our team evaluates custom integrations for every implementation.
Start with API manufacturing software consultation
Understand how ERPNext manages compliance, yields, and complex processes. Sigzen helps you evaluate what fits your operations before you commit.
Frequently Asked Questions
Which is the best ERP software for Active Pharmaceutical Ingredients businesses in India?
ERP software for manufacturing is a centralized system like SigzenMFG that integrates production, inventory, finance, and supply chain processes. It streamlines operations by providing real-time data visibility and automation. This reduces manual effort and improves coordination across departments. In practice, it enhances efficiency, reduces operational costs, and enables better decision-making across manufacturing operations.
How long does ERP implementation take for a Active Pharmaceutical Ingredients company?
Implementation timelines typically range from 8 to 16 weeks depending on process complexity and data readiness. API manufacturing software setups require mapping production stages, compliance workflows, and integrations. A structured rollout ensures minimal disruption and faster adoption.
Can this ERP migrate data from Tally for a Active Pharmaceutical Ingredients business?
Yes, ERPNext supports migration of financial and basic operational data from Tally. Historical transactions, ledgers, and balances can be imported. This helps API ERP users move away from disconnected systems without losing important records.
Does Sigzen Technologies ERP support compliance for Active Pharmaceutical Ingredients manufacturers?
Yes, the system is configured to support GMP, CFR Part 11, and audit trail requirements. It helps maintain proper documentation, batch traceability, and controlled access. This makes it suitable for global pharma compliance needs in API manufacturing.
How does ERP handle CFR Part 11 compliance in API manufacturing?
ERPNext maintains secure electronic records with audit trails and user authentication. Every action is tracked with timestamps, ensuring data integrity. This helps API manufacturing software meet regulatory expectations without manual record-keeping risks.
How can ERP improve yield tracking in API production?
ERP systems track yields at each production stage instead of only final output. This helps identify losses early and improve efficiency. With API ERP tools, production teams gain better control over materials and process outcomes.
Is ERP worth it for small API manufacturers in India?
Yes, especially if you face compliance pressure or operational inefficiencies. Even smaller API units benefit from structured processes and traceability. If you want to see how it fits your setup, book a demo and evaluate it with real scenarios.