Serialized Medical Software becomes essential when serial tracking gaps, incomplete device history records, and delayed documentation slow your operations. With ERPNext, you gain full traceability, structured compliance workflows, and reliable reporting. Delivered as an ERPNext-based Medical Devices & Diagnostics ERP solution by Sigzen, it gives you the confidence to manage audits and operations without uncertainty.

Medical device ERP challenges in tracking and compliance
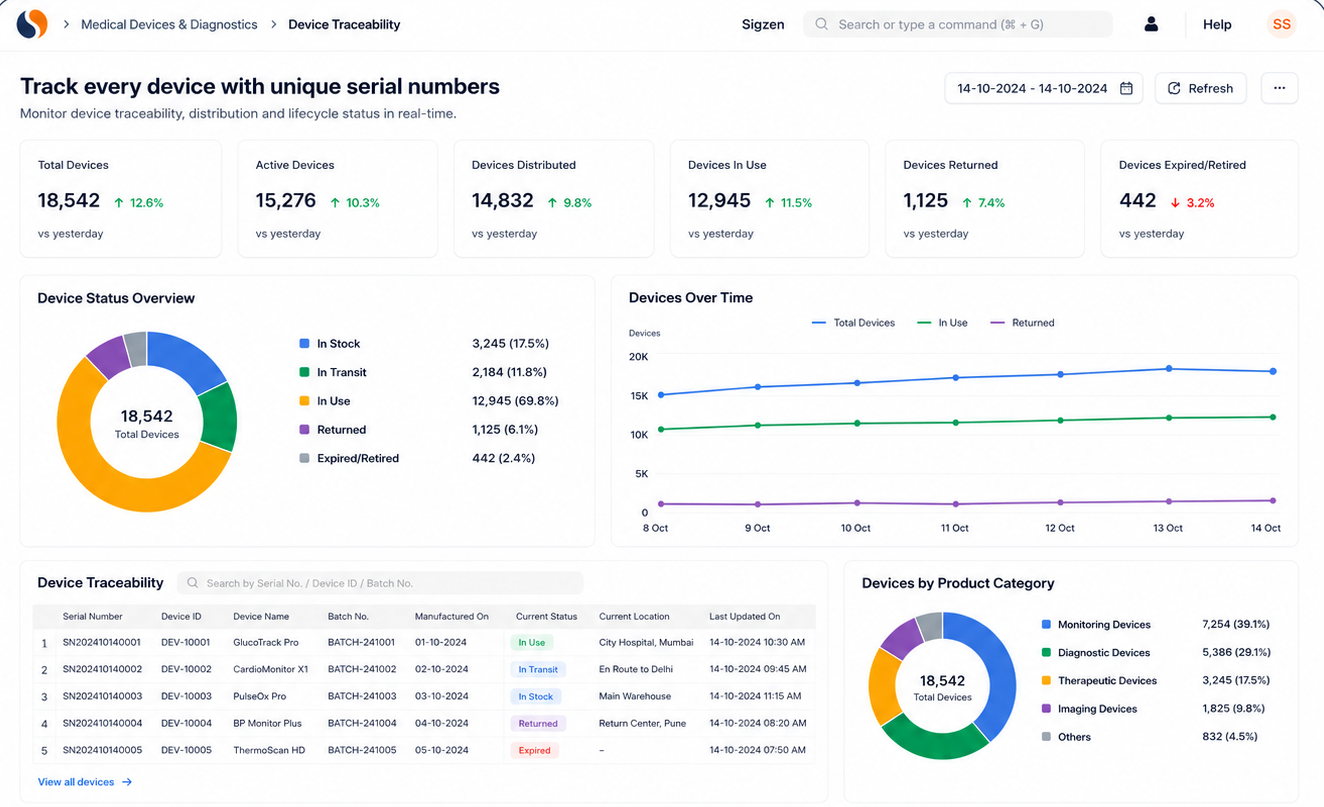
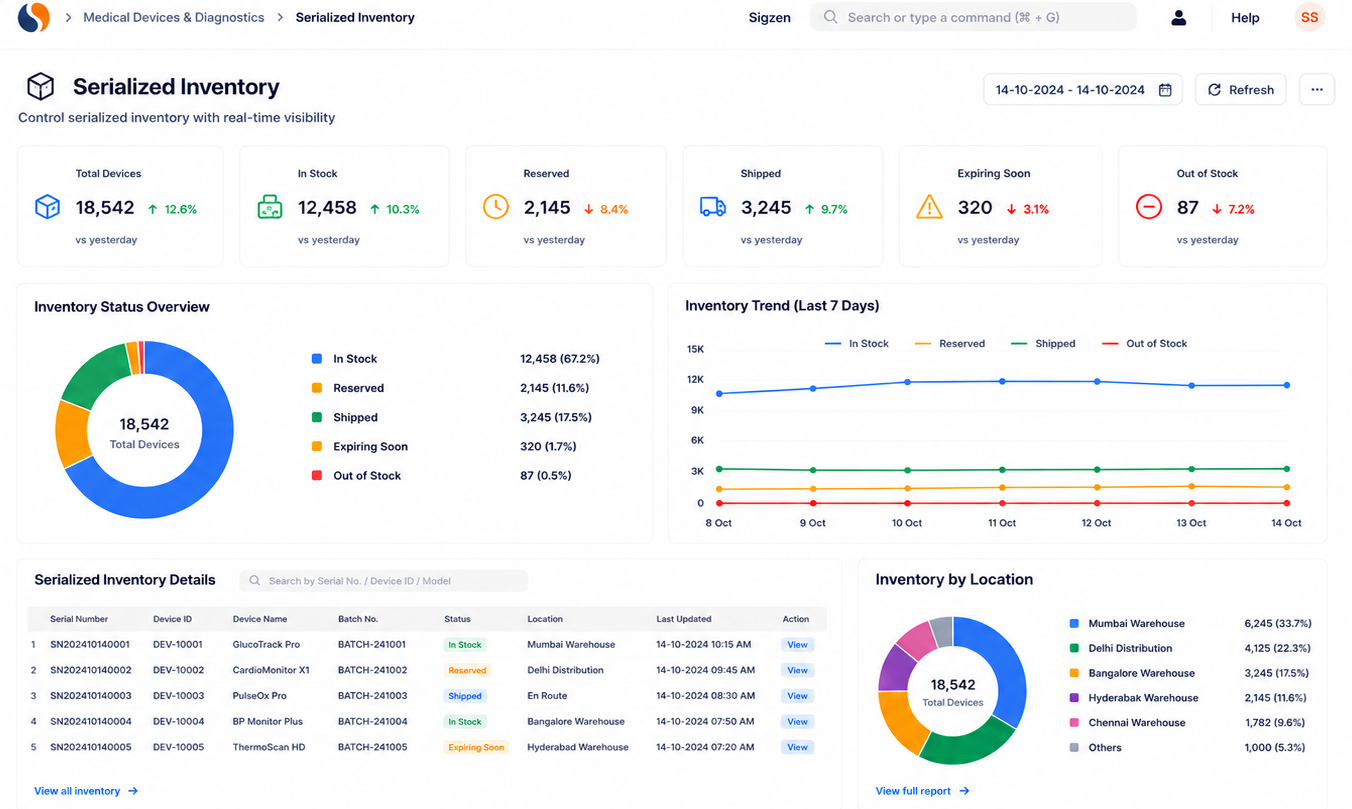
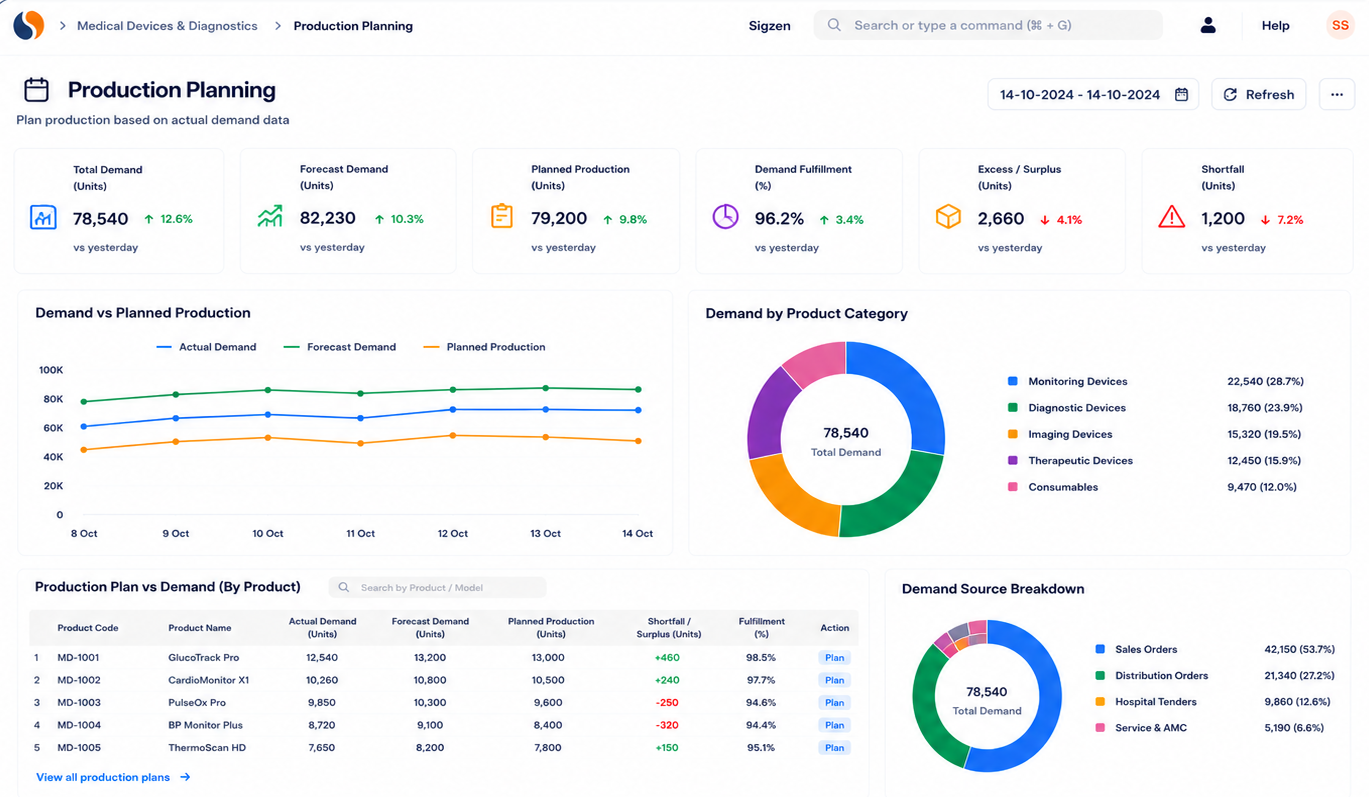
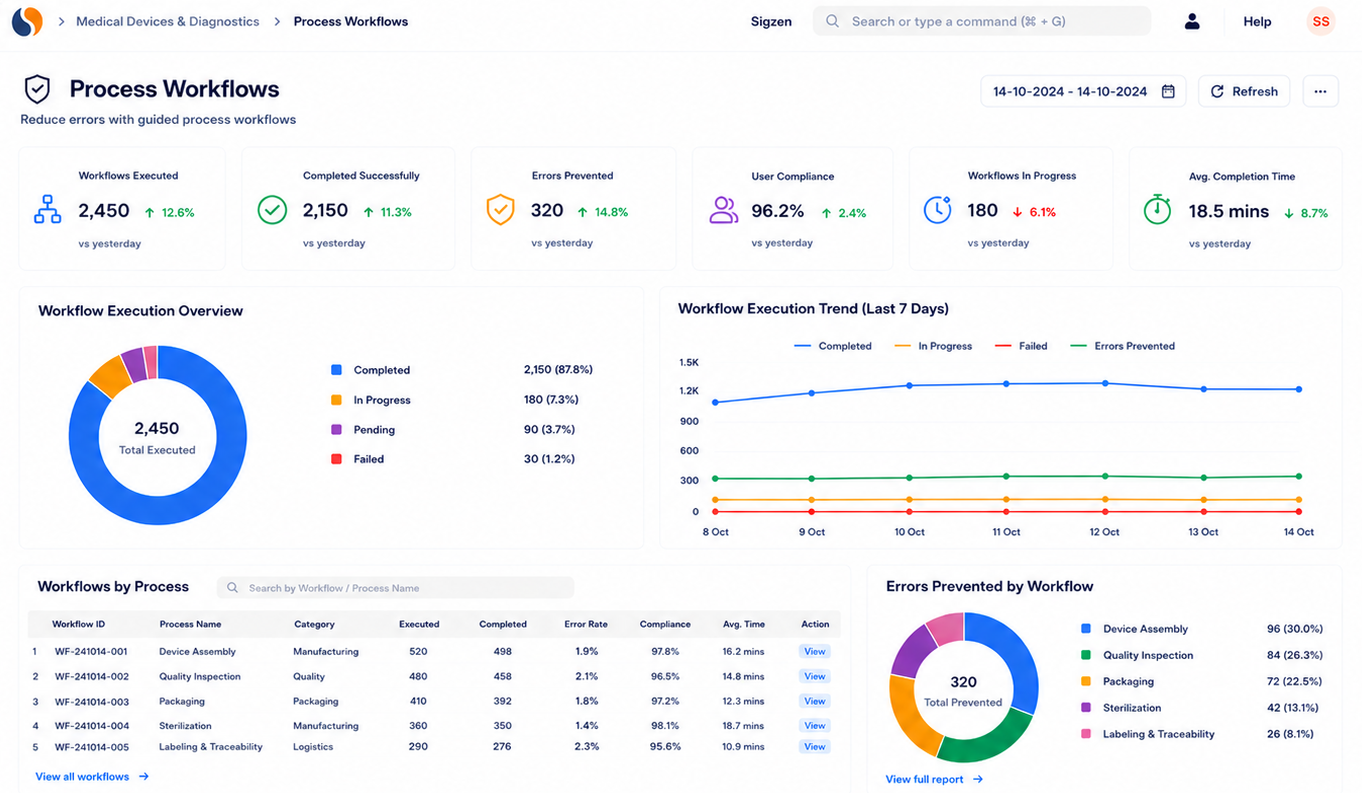
Key features of medical device ERP for serialized tracking
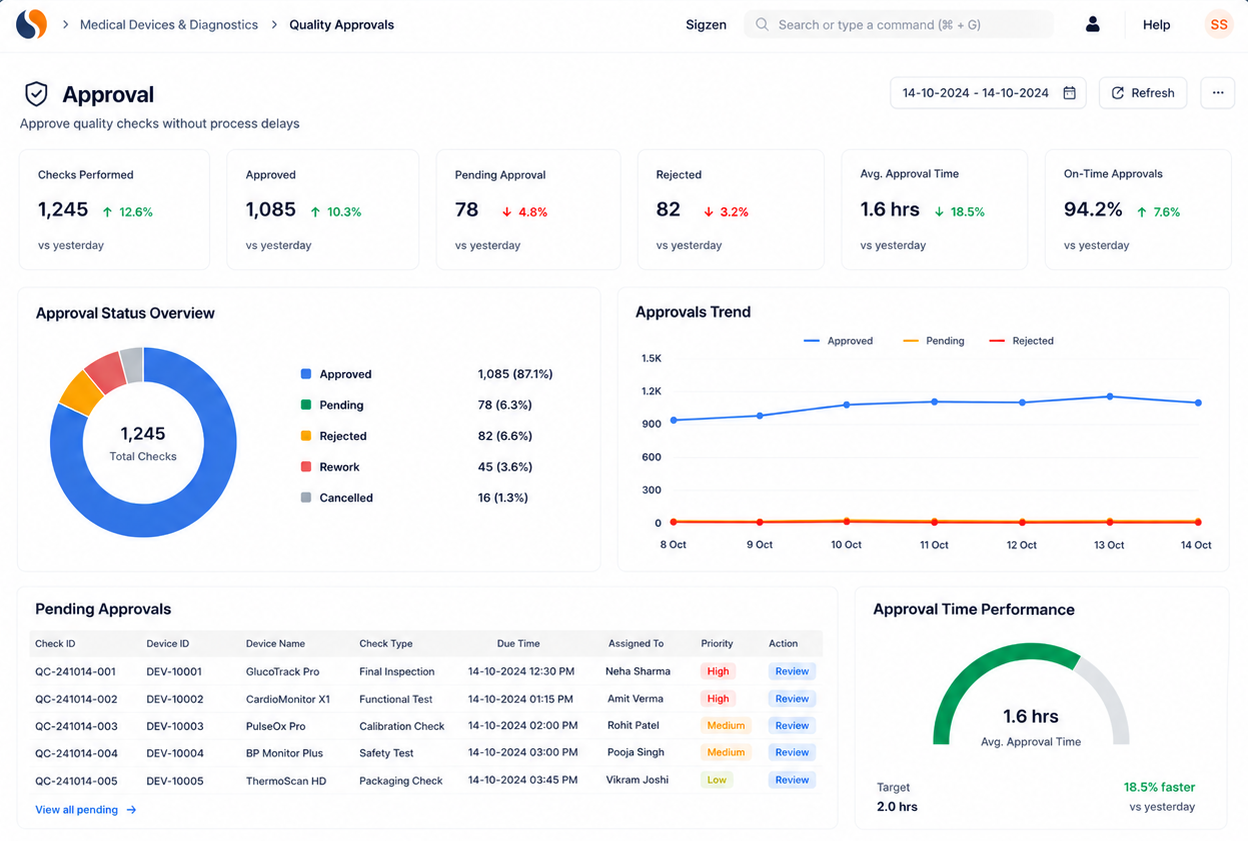
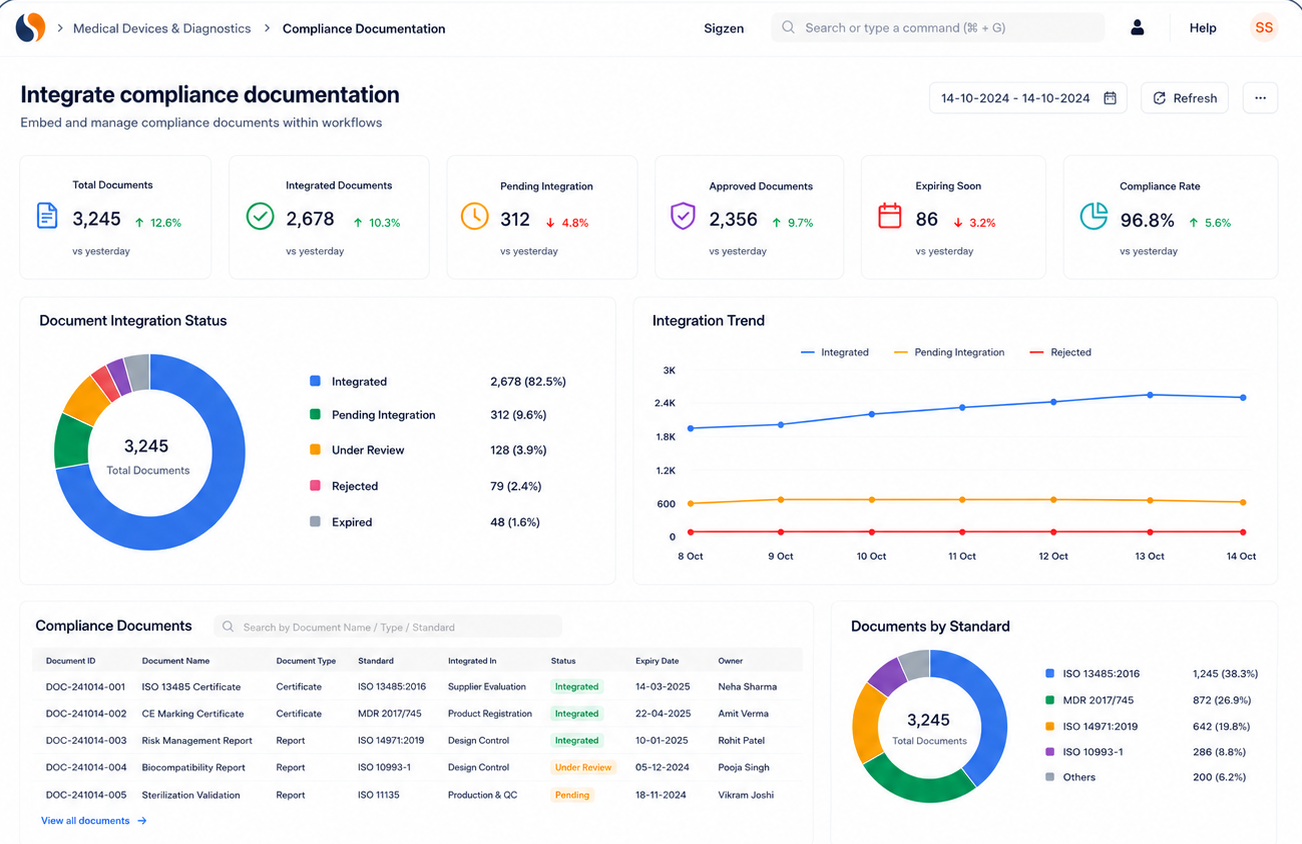
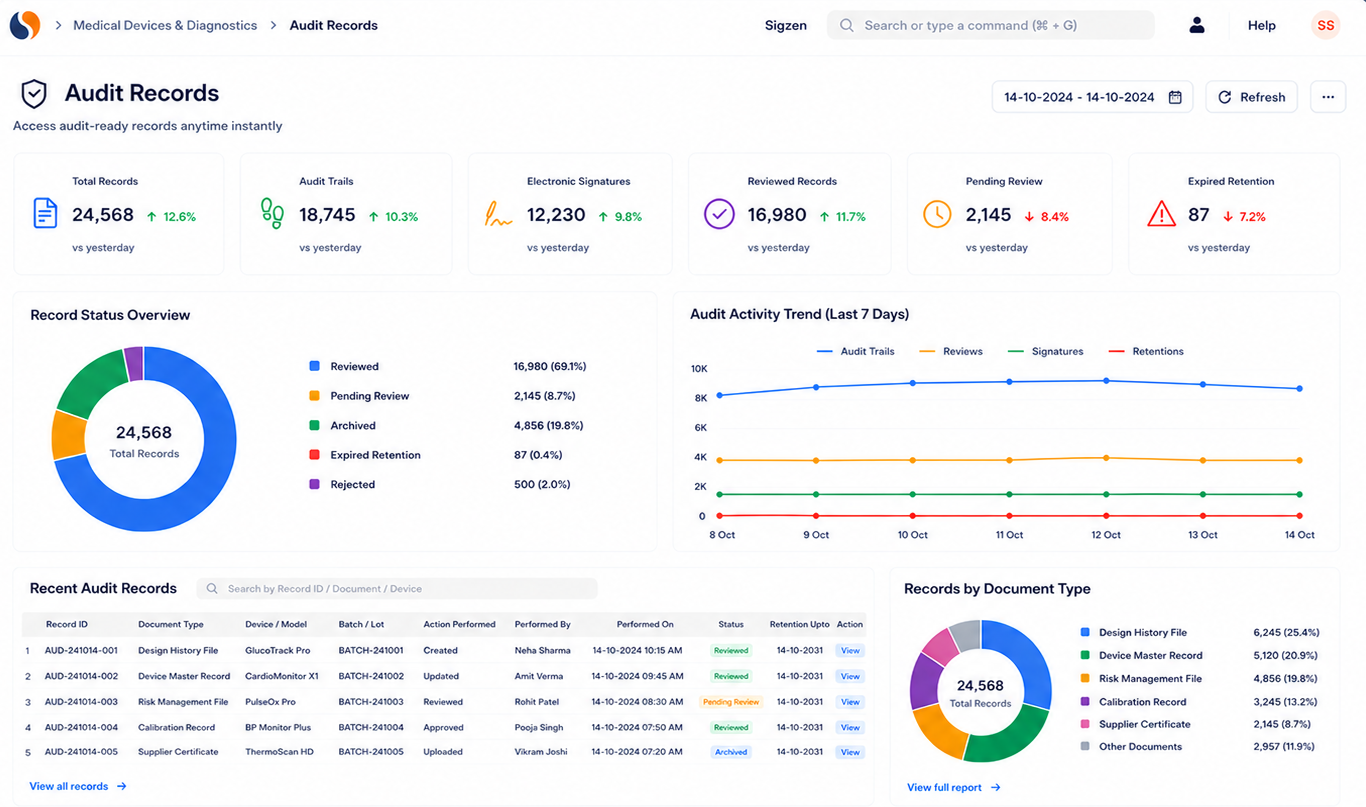
Built for compliance and audit readiness
Works with the tools your team already uses
Medical device operations involve multiple systems across production, quality, and finance. Integrations ensure your data flows without duplication or errors.
You continue using your existing tools while ERPNext connects them into one structured workflow.
Already using a tool not listed here? Our team evaluates custom integrations for every implementation.
Improve Device Traceability and Process Accuracy
Manage serial tracking, device history records, and compliance workflows with ERPNext tailored to your operations.
Explore Medical Device ERP Solution
Frequently asked questions about Medical Devices ERP
Which is the best ERP software for Medical Devices businesses in India?
The best ERP software for medical devices should support serial tracking, compliance, and documentation. Medical Device ERP, ISO 13485 Compliance, Serialized Medical Software built on ERPNext helps manage production, quality, and inventory in one system. It suits manufacturers who need full traceability and control.
How long does ERP implementation take for a Medical Devices company?
ERP implementation for medical devices typically takes 8–16 weeks depending on complexity. It includes setup, migration, testing, and training. ERPNext focuses on structured workflows like serialized tracking and compliance documentation for faster adoption.
How does ERPNext support data migration for medical device manufacturers?
ERPNext supports migration of item masters, serial numbers, device history records, inventory data, and compliance documents into the Medical Device ERP system. This ensures continuity while improving process visibility. It helps maintain accurate records required for ISO 13485 compliance and audit readiness.
Does Sigzen Technologies ERP support compliance for Medical Devices manufacturers?
Yes, ERPNext supports ISO 13485 compliance, audit trails, and regulatory documentation. It ensures that all records are properly maintained for inspections. This Medical Device ERP setup helps manage compliance as part of daily operations.
How do medical device companies manage serial tracking effectively?
Serial tracking involves assigning unique identifiers to each product. ERPNext tracks these identifiers across production, testing, and dispatch. This improves traceability and supports compliance requirements.
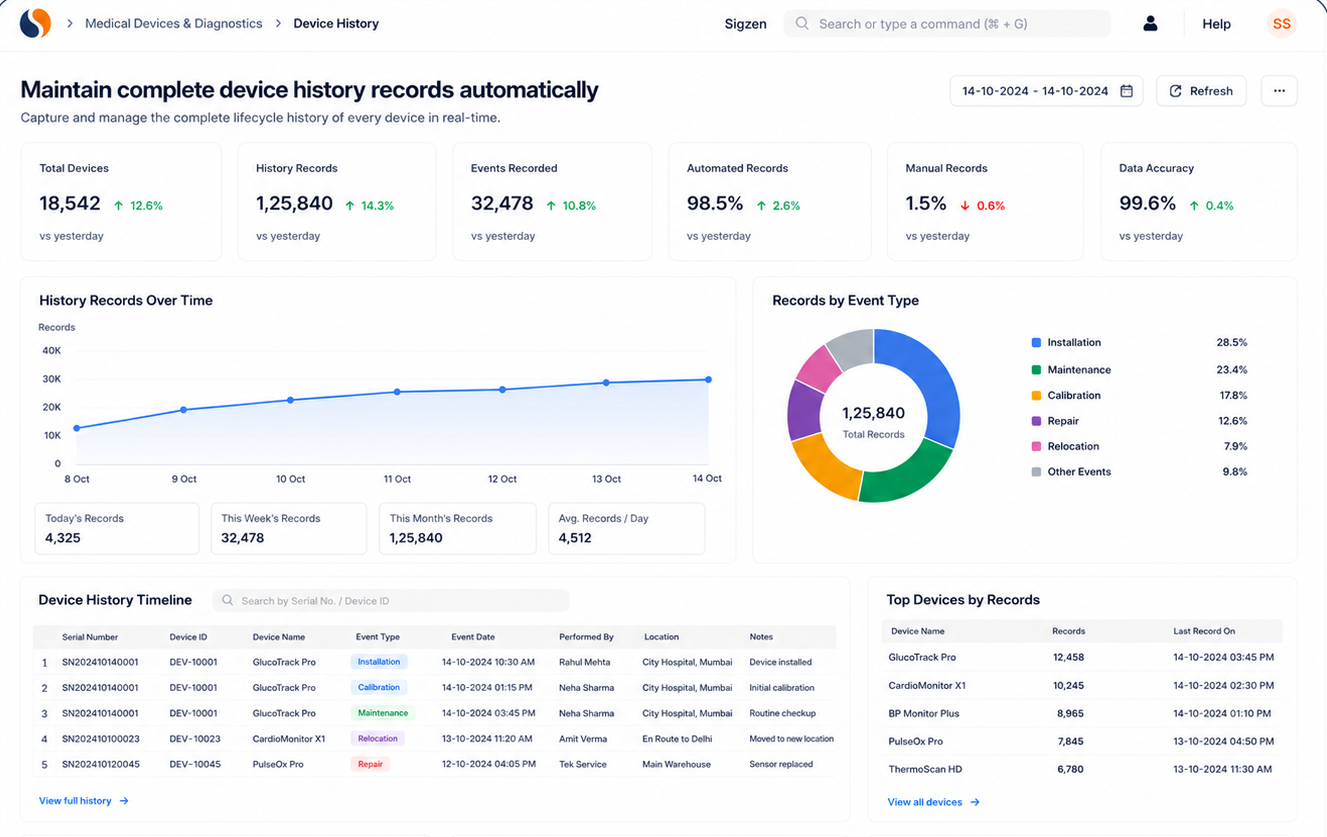
Why do device history records become incomplete?
Incomplete records often result from manual entries and disconnected systems. Data gets missed or duplicated across departments. A structured ERP system ensures every step is recorded accurately.
Is ERP necessary for small medical device manufacturers?
Yes, even small manufacturers benefit from ERP when managing compliance and tracking. ERPNext helps control processes and reduce errors. If you want better visibility and fewer risks, you can explore this system further.