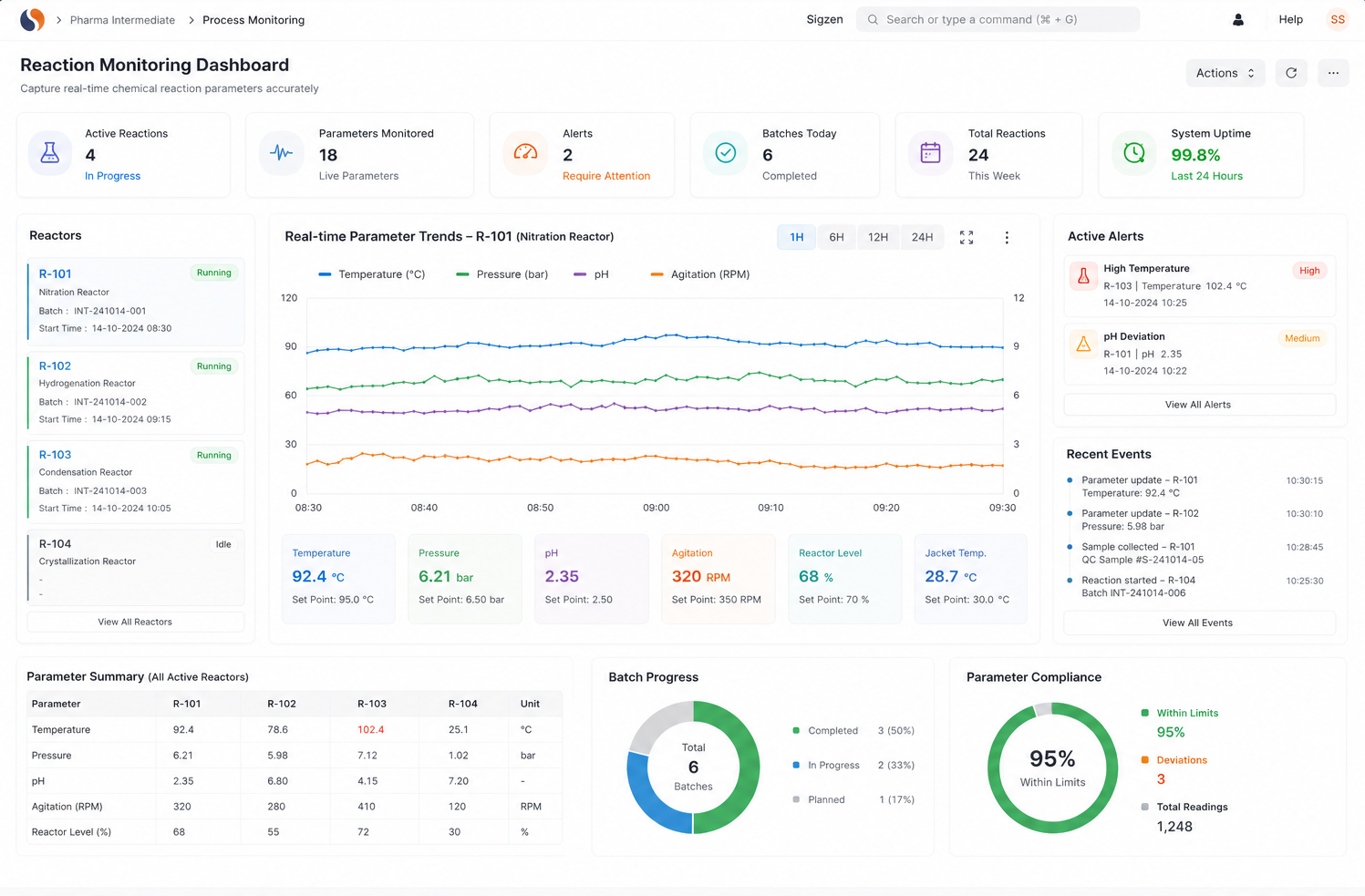
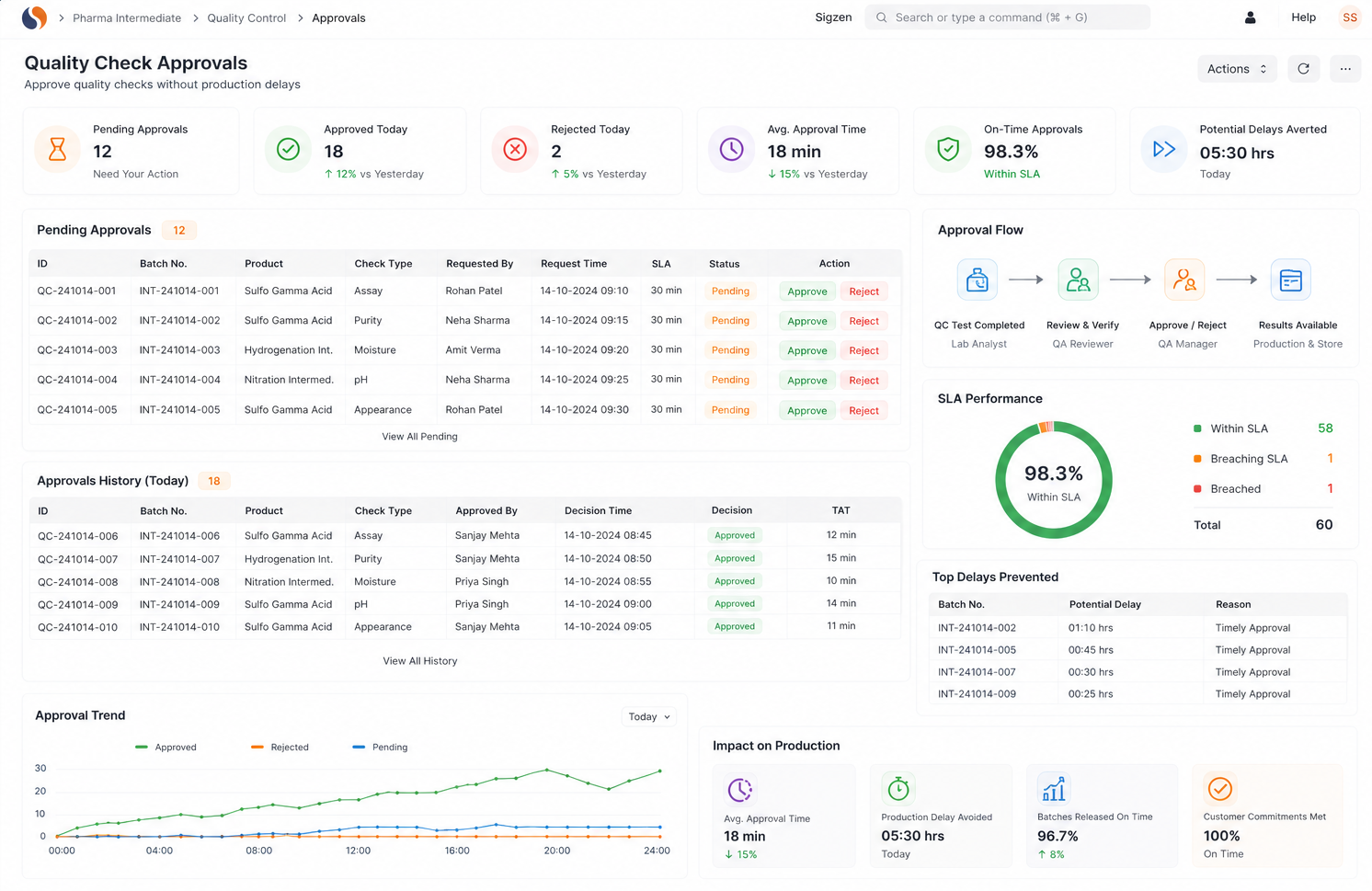
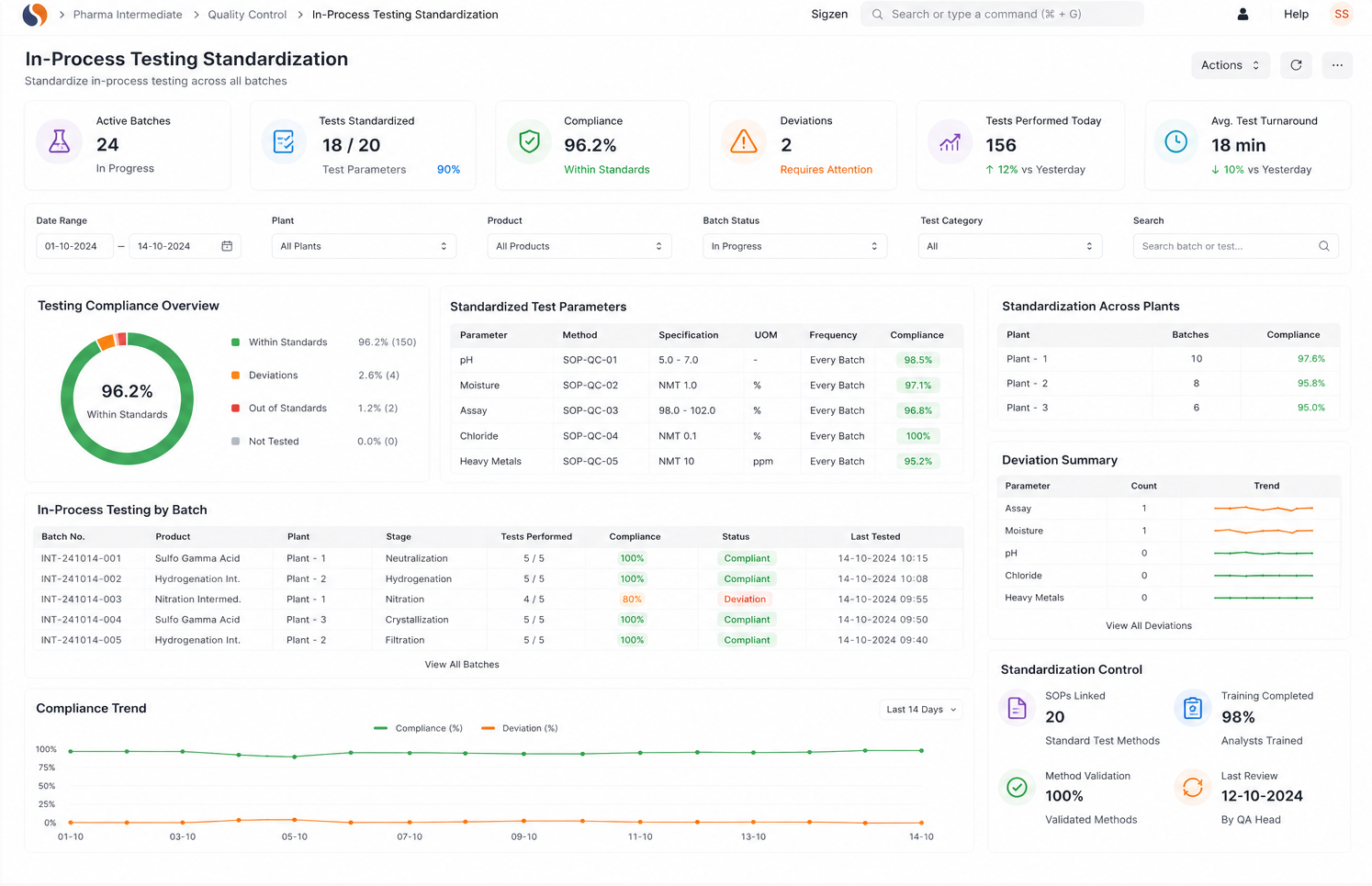
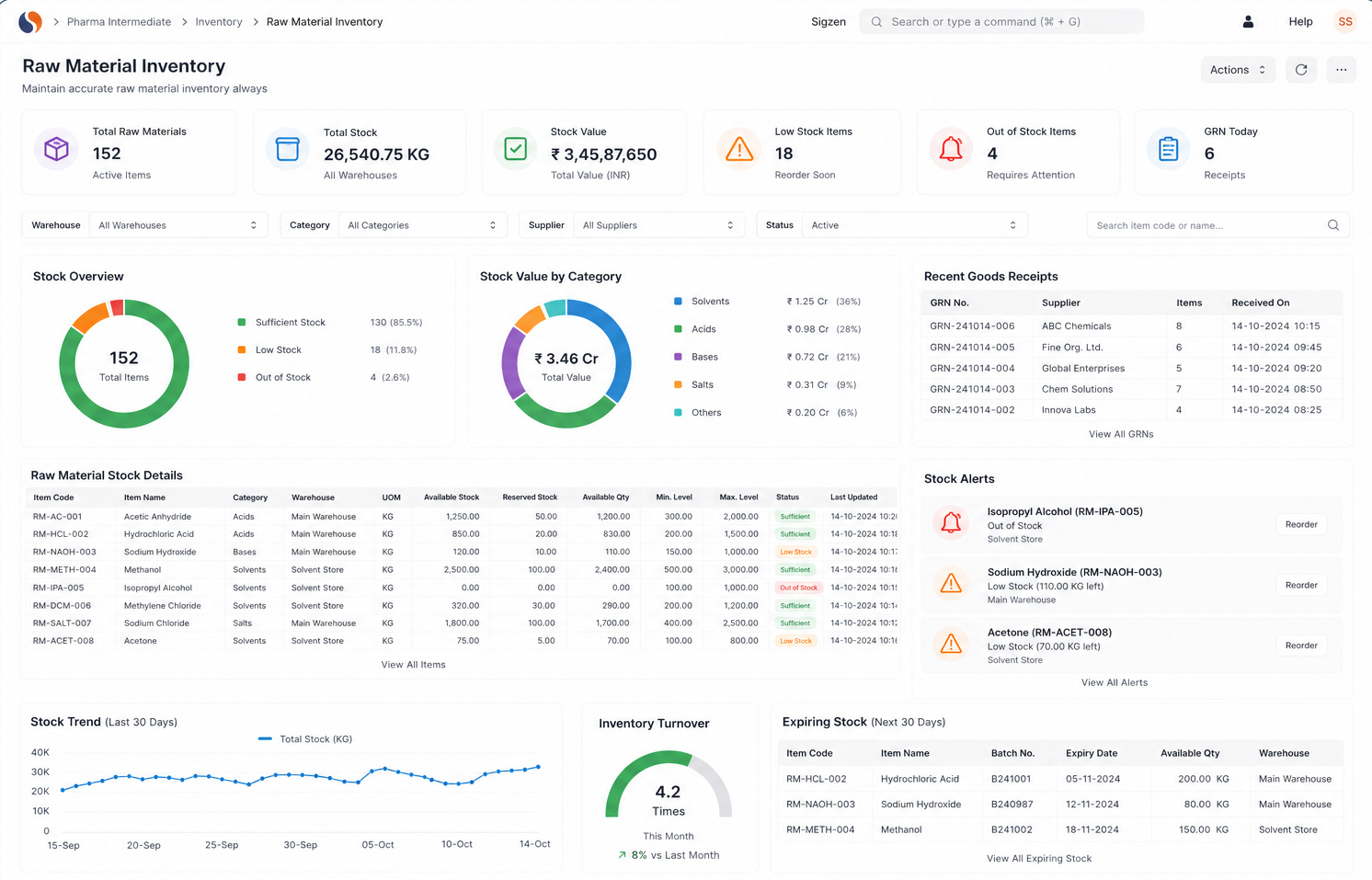
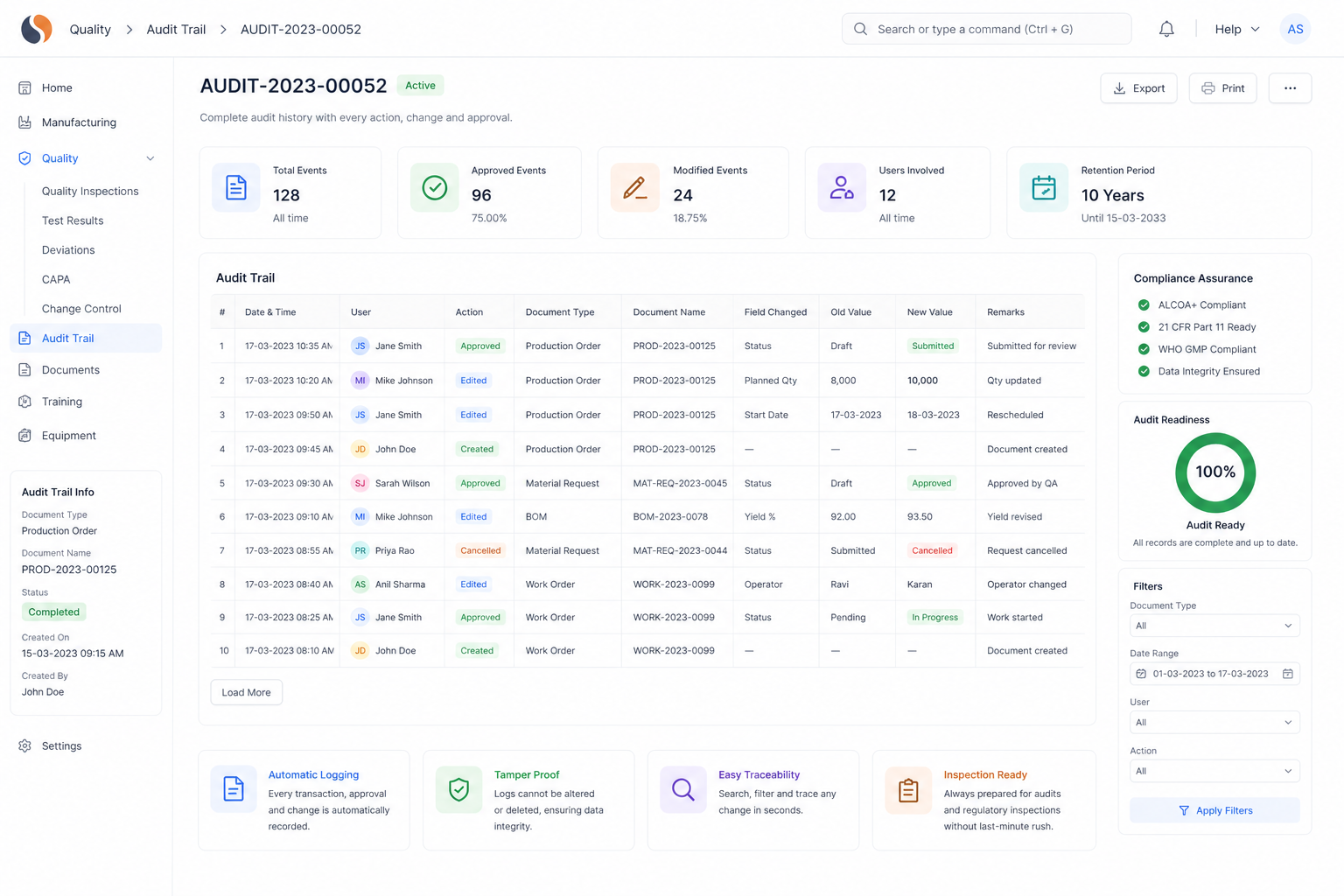
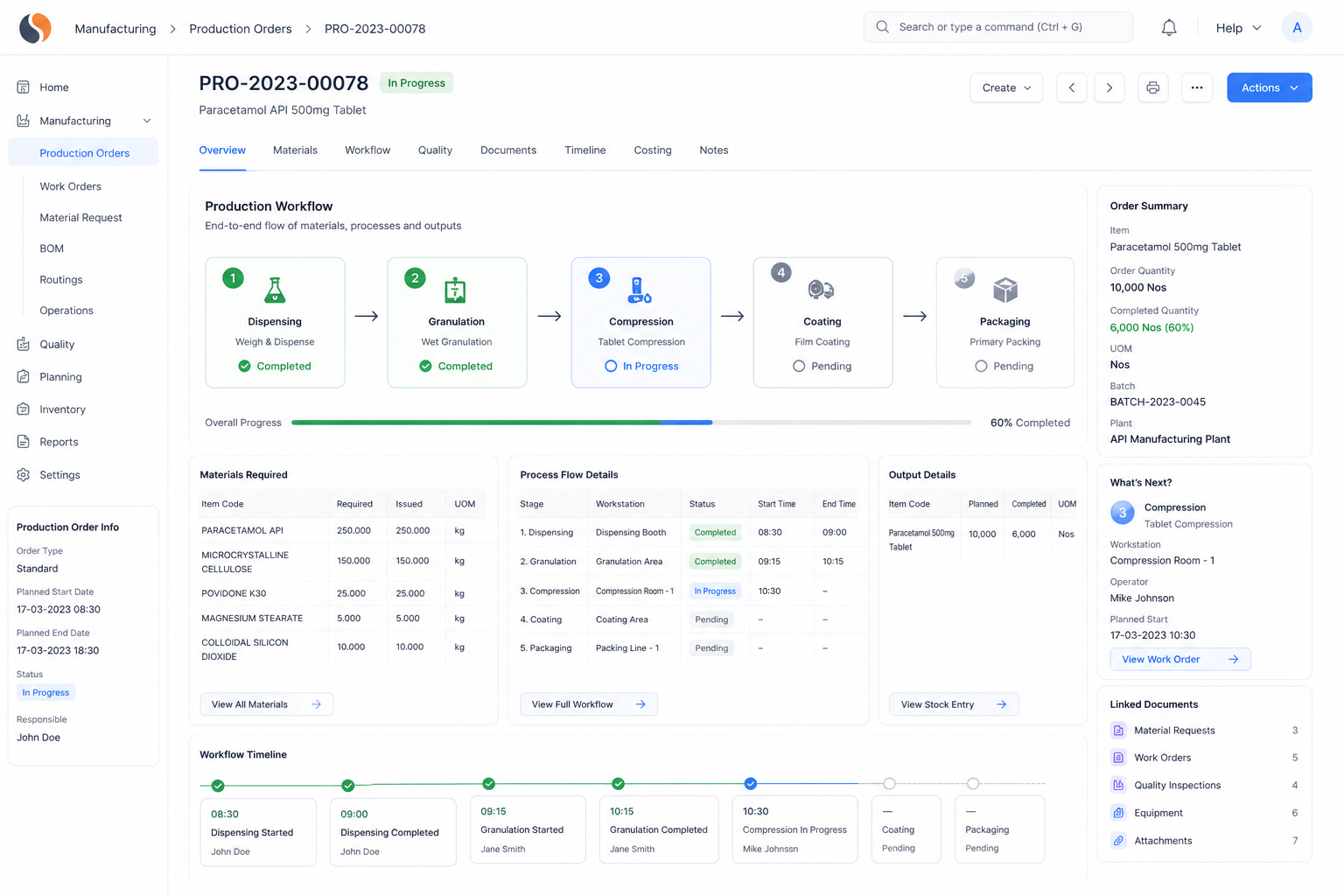
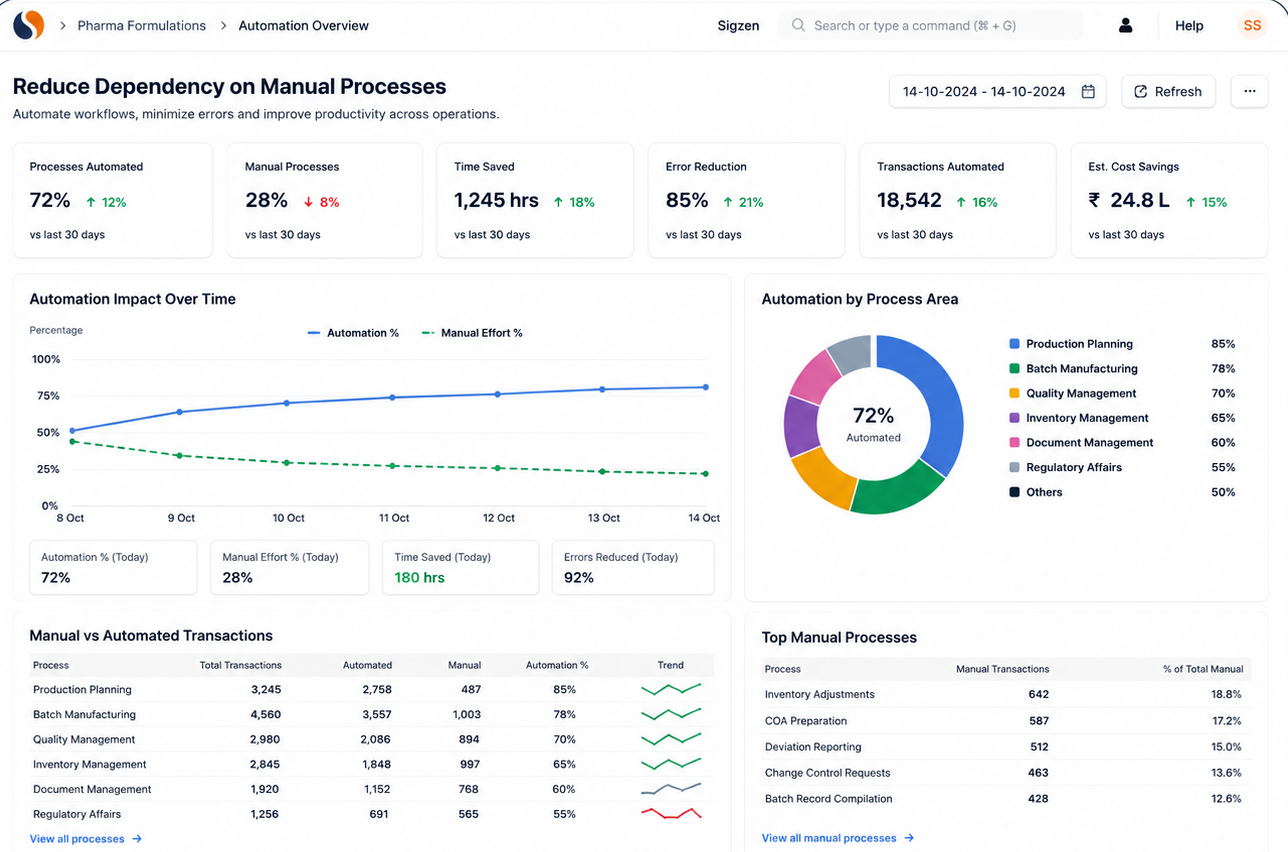
Chemical Reaction Tracking is critical when batch errors, delayed quality checks, and manual records slow your operations. With ERPNext based Pharma Intermediate Erp Solution implemented by Sigzen Technologies, you gain clear batch visibility and controlled processes. You see what’s happening in real time and trust the numbers you rely on.

Challenges in Pharma Intermediate ERP
Key Features of Pharma Intermediate ERP and Reaction Tracking
Built for compliance and audit readiness
Works with the tools your team already uses
Pharma intermediate manufacturing involves multiple systems across production, quality, and finance. Integrations ensure your data flows without duplication or manual work.
You continue using familiar tools while ERPNext connects them into one workflow.
Already using a tool not listed here? Our team evaluates custom integrations for every implementation.
Get Pharma Intermediate ERP for Better Batch Control
Simplify batch traceability, quality checks, and reporting with ERPNext tailored to your operations.
Frequently asked questions about Pharma Intermediates ERP
Which is the best ERP software for Pharma Intermediates businesses in India?
The best ERP software for pharma intermediates businesses in India is one that supports batch tracking, quality control, and compliance. ERPNext with Pharma Intermediate ERP, Chemical Reaction Tracking helps manage production, testing, and inventory in one system. It suits manufacturers who need visibility and control without complex systems.
How long does ERP implementation take for a Pharma Intermediates company?
ERP implementation for pharma intermediates typically takes 8–16 weeks depending on process complexity. It includes setup, data migration, testing, and training. With ERPNext, the focus is on practical workflows like batch tracking and chemical reaction tracking, ensuring faster adoption and minimal disruption.
How does ERPNext support data migration for Pharma Intermediates?
ERPNext supports data migration for item masters, stock balances, supplier records, customer details, batch history, and quality records. For pharma intermediate manufacturers, this ensures a smoother shift to digital production control. It helps maintain continuity while improving batch tracking, reaction monitoring, and compliance documentation.
Does Sigzen Technologies ERP support compliance for Pharma Intermediates manufacturers?
Yes, ERPNext configured by Sigzen Technologies supports GMP guidelines, GST compliance, and audit trails. It ensures proper documentation for batch traceability and quality control. This Pharma Intermediate ERP, Chemical Reaction Tracking setup helps you stay compliant without manual effort.
How do I track chemical reactions in ERP software?
Chemical reaction tracking in ERP involves recording parameters like temperature, pressure, and duration during production. ERPNext allows operators to enter this data in real time, linking it to batch records. This improves accuracy and ensures reliable process documentation.
Why do pharma intermediate companies struggle with batch traceability?
Most companies rely on manual records or disconnected systems, making it hard to link raw materials to finished batches. This leads to delays during audits and investigations. A Pharma Intermediate ERP system solves this by maintaining complete batch traceability automatically.
Is ERP necessary for small pharma intermediate manufacturers?
Yes, even small manufacturers benefit from ERP when managing batch production and compliance. ERPNext helps control inventory, track reactions, and maintain quality records without increasing complexity. If you want better visibility and fewer errors, it’s worth considering a demo.